What is Battery Efficiency?
When selecting a battery for material handling equipment, busy fleet managers are going to ask one question: how energy efficient is the battery? In other words, how much bang can we get for our buck?
Battery efficiency, simply put, is the amount of energy you can get out of a battery relative to the amount of energy that’s put into it.
The amount of energy you get out is always going to be less than what’s put in - however, there are specific factors that will affect that difference.
Coulombic Efficiency and Voltage Efficiency
A battery’s energy efficiency can be measured in two ways: coulombic efficiency (CE) and voltage efficiency.
CE measures the transfer of electrons during charge and discharge as well as how many electrons are lost during a full cycle. A coulomb counter is often used to make these measurements. The higher the CE, the lower the electron loss, and the longer the battery life.
Voltage efficiency is the voltage difference when a battery is charging versus discharging - this difference is caused by what’s known as over-potential.
Every rechargeable battery must be charged at a higher voltage than the discharge voltage - this difference is key to determining how efficient the battery is.
Five key factors affect industrial battery efficiency, and the two methods of measurement described above.
- Charge Current
- State of Charge
- Internal Resistance
- Battery Temperature
- Battery Age
1: Charge Current
For lithium-ion batteries, charging 'best practice' is to keep the current controlled at a moderate level to maximize the battery’s efficiency and lifespan.
Here’s why: during the charging process, changes occur within the battery’s internal chemistry, and charging at a high current worsens these effects.
Lithium atoms and electrolytes build up on the surface of the graphite anode, forming a layer called solid electrolyte interface (SEI) which protects the anode, but also grows thicker over time and can obstruct ion access to the anode if too thick.
On the cathode, a similar build-up of lithium ions forms, which can cause electrolyte oxidation and lead to thermal runaway.
When the charge current is too low, it improves battery longevity but reduces the battery’s capacity. It is also time inefficient.
Lithium-ion batteries are often designed for charging in as little as an hour, as the efficiency loss is sometimes less important than time loss.
2: State of Charge
The battery state of charge for an electric vehicle like a forklift is the equivalent of the fuel gauge - it’s the level of charge relative to its capacity at any given moment.
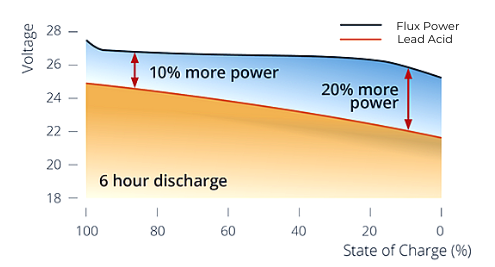
Throughout the discharge cycle, the voltage output gradually drops as the SoC declines as well. Lithium-ion batteries have a much lower rate of voltage decline than lead acid batteries.
The capacity loss that batteries experience when they’re cycled at high temperatures is directly related to their SoC - the higher the SoC, the worse the capacity loss is.
It’s important to be cognizant of a battery’s SoC to maximize its efficiency. Due to the flat discharge curve, a coulomb counter is often the only way to accurately measure and track SoC for lithium-ion. Most systems simply measure the output voltage to estimate SoC, but this method can be inaccurate and is affected by temperature, surface charge, and age.
3: Internal Resistance
A battery’s internal resistance is affected by many factors including size, age, current, and chemistry. The lower the internal resistance, the easier it is for the battery to perform. Lithium-ion batteries have one of the lowest internal resistances available.
In lithium-ion batteries, the SEI on the anode contributes to a high internal resistance by blocking interaction with the graphite.
The SEI layer is essential to the battery’s functionality because it stabilizes the system and increases lifespan, but its effects can increase internal resistance over time.
Lithium-ion battery manufacturers use additives to the battery’s electrolyte to reduce some of this effect and prevent the SEI film from becoming too restrictive.
Most manufacturers keep their additive formula a secret, but vinylene carbonate is a common one that is highly effective at keeping the internal resistance low with use and age.
4: Battery Temperature
Lithium-ion batteries should be charged at a range of 32° F through 113° F, and discharged between -4° F through 131° F. Their charge and discharge performance remain good at higher temperatures compared to other batteries, but the more they’re exposed to high temperatures, the shorter their lifespan.
When the temperature is below 41° F, the charge current should be reduced.
High temperatures cause cathode electrolyte oxidation, which can result in a sudden loss of capacity.
Charging a lithium-ion battery at temperatures below freezing will cause permanent SEI build-up on the anode, which damages the battery and reduces capacity.
5: Battery Age
It seems obvious that the more a battery ages, the less efficient it is - but battery age isn’t just counted in years.
Generally, lithium-ion batteries last for 2,000-3,000 cycles, which is significantly longer than a lead acid battery’s lifespan of 1,000-1,500 cycles.
What does battery aging look like? The battery will experience an irreversible loss of capacity. Liquid electrolytes will slowly dry up or turn solid, and a layer of lithium ions will build upon the electrodes.
Overcharging, deep cycling, and extreme temperatures will all speed up the aging process for a lithium-ion battery. To maximize a lithium-ion battery’s lifespan, it’s best to opportunity charge at moderate (room) temperatures.
What’s the most efficient battery for forklifts?
Lithium-ion batteries have one of the highest CE ratings out of all types of rechargeable batteries at 99% or higher. These are the most efficient batteries. Lead acid batteries are lower at about 90%, and nickel-based batteries are closer to 80%. These efficiencies drop at high charge rates. Lithium-ion stays close to 90% at a 1C charge rate, while lead acid drops below 50% efficiency.
Lithium-ion batteries:
- Have a longer lifespan both in number of cycles and capacity retention over time than lead acid batteries
- Can be partially charged over and over again while lead acid requires a weekly overcharge/equalization to balance cells and remove sulfation
- Maintain high voltage output at a lower state of charge while lead acid batteries decline in voltage as their SoC declines
- Can operate at higher temperatures compared to lead acid, and lower temperatures with built-in heaters
If you are trying to get the most out of your material handling equipment, lithium-ion batteries will be the most efficient solution for multi-shift operations. Because lithium-ion batteries can last for up to 3 shifts versus lead acid batteries lasting only 1 to 2 shifts, the decision to switch to lithium-ion technology is an easier one.












